Clinical utilization of four-factor prothrombin complex concentrate: a retrospective single center study
Article information
Abstract
Objective
Four-factor prothrombin complex concentrate (4F-PCC) was approved by the US Food and Drug Administration in 2013 for management of severely bleeding patients on warfarin therapy. We describe use of 4F-PCC at a large, suburban academic center.
Methods
We retrospectively reviewed all patients receiving 4F-PCC from its introduction through 2016 at a large level 1 trauma center. Clinical and demographic data were obtained, including indications for anticoagulation and antiplatelet agents, comorbidities, concomitant medications, etiology and site of bleeding, as well as disposition, length of stay, mortality, and thrombotic events.
Results
One hundred eighty-four patients received 4F-PCC. Mean age was 72 years; 40.8% were female. Indications for 4F-PCC administration included: active bleeding (74%), reversal prior to a procedure (14%), and elevated international normalized ratio (12%). Warfarin was the most common concomitant medication (71.1%). Most patients were receiving anticoagulation for atrial fibrillation (63%). Concomitant treatments for bleeding included vitamin K (58.2%), packed red blood cells (50%), fresh frozen plasma (38%), and platelets (26.1%), amongst others. Median length of hospital stay was 8.4 days. Nine patients (4.9%) developed thrombosis within 90 days of 4F-PCC. Mortality was 24.5%, with notably higher rates amongst those who received 4F-PCC for off-label indications (19.1% on-label mortality vs. 37.7% off-label mortality on chi-square analysis, P=0.01).
Conclusion
This study demonstrates that 4F-PCC is being utilized for indications other than the reversal of warfarin-induced coagulopathy. Further investigation is warranted to determine the efficacy and safety of 4F-PCC for these potential indications.
INTRODUCTION
Warfarin, an oral anticoagulant, is commonly used for the prevention or treatment of thrombotic and embolic conditions, including atrial fibrillation, pulmonary embolism, and deep venous thrombosis [1]. Warfarin is a vitamin K antagonist with a narrow therapeutic window and variable individual-specific dosing, and as such, carries a significant risk of bleeding, with reported rates ranging from 0% to 10% [1-3]. Reversal of warfarin-associated bleeding has traditionally been accomplished using vitamin K or fresh frozen plasma (FFP) due to the widespread availability and relatively low cost of these agents [4]. Disadvantages to using FFP include the need for ABO compatibility testing prior to administration, large volume requirements of up to 10–15 mL/kg, and prolonged thawing time [4-6].
For these reasons, recombinant vitamin K-dependent factors and 3- or 4-factor prothrombin complex concentrates (4F-PCCs) have been increasingly utilized on or off-label for the reversal of warfarin-associated bleeding. 4F-PCC (Kcentra; CSL Behring, King of Prussia, PA, USA), which gained US Food and Drug Administration approval in 2013, is a blood coagulation factor replacement product containing the vitamin-K dependent coagulation factors II, VII, IX, X, proteins C and S, antithrombin III, and human albumin [7,8]. It is the only drug to be approved to date for the rapid reversal of warfarin in the setting of acute major bleeding or urgent need for surgical or invasive procedures [7].
Despite its rising popularity, few studies have reported indications and outcomes with use of 4F-PCC, and those that do focus on niche patient populations or specific dosing regimens [9-11]. In our study, we describe use of 4F-PCC in the emergency department at a large, suburban academic center to elucidate practice patterns.
METHODS
Study design
We conducted a structured retrospective chart review of all adult patients receiving 4F-PCC from when it became available to our hospital in 2015 through 2016. Data were obtained from electronic medical records reviewed in Cerner PowerChart, the centralized clinical database for our hospital. Three independent reviewers extracted data to ensure reliable capturing; any discrepancies were resolved via discussion amongst the reviewers. Medical record review was consistent with the recommendations of Gilbert et al. [12], as well as that of Kaji et al. [13]. Our study also followed the Strengthening of Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines for cross-sectional studies (http://www.equator-network.org/reporting-guidelines/strobe/). Because of its retrospective design, this study was approved by our institutional review board (2017-4079) with waiver of informed consent.
Setting
The study was conducted at the emergency department of a university affiliated, tertiary care, suburban hospital in the northeast region of the US with an annual census of approximately 110,000 patients.
Data measures
Demographic and clinical data points of interest included indications for anticoagulation at baseline, concomitant anti-thrombotic use (agents of interest were warfarin, aspirin, clopidogrel, and direct oral anticoagulants), presence of bleed (and if present, bleed type and site of bleeding), and indications for reversal of anticoagulation with 4F-PCC. 4F-PCC use was considered “on-label” if it was administered for warfarin reversal in patients with either acute major bleeding or requiring urgent surgery/invasive procedure [7]. Any use of 4F-PCC outside this scope was deemed off-label.
Study outcomes
The primary outcome of interest was the incidence of mortality, defined as failure to survive to hospital discharge, in patients receiving 4F-PCC for an on-label versus off-label indication. Secondary outcomes included patient disposition, length of hospital stay, and subsequent thrombotic event within 90 days of 4F-PCC administration. A 90-day assessment period was chosen in line with the cut-off designated by prior studies to facilitate capturing of long-term sequelae of reversal [6].
Data analysis
Data were input into the REDCap electronic data capture system and summarized with descriptive and comparative statistics. Categorical data were summarized as frequencies and percentages. Continuous data were summarized as means and standard deviations. Patient subgroups were compared using chi-square statistics. The level of significance was set as a P-value of <0.05.
RESULTS
One hundred eighty-four patients received 4F-PCC during the study period. Mean±standard deviation age was 72±17 years; 40.8% were female. Of all patients, 73.9% presented to the emergency department with a bleed, of which 36.8% were traumatic in etiology. The most common bleeding sites were intracranial (28.8%), lower gastrointestinal (15.2%), upper gastrointestinal (10.3%), hemothorax (3.3%), abdomen (3.3%), retroperitoneal (3.8%), extremities (22%), pelvic (2.2%), and epistaxis (2.2%). The remaining 48 patients presenting with no bleed required reversal for an upcoming procedure (52.1%) or for an elevated international normalized ratio (INR, 47.9%). For patients with traumatic events, the most common bleeding sites were intracranial (60%), hemothorax (12%), and abdominal bleeds (10%). Indications for 4F-PCC administration are depicted pictorially in Fig. 1.
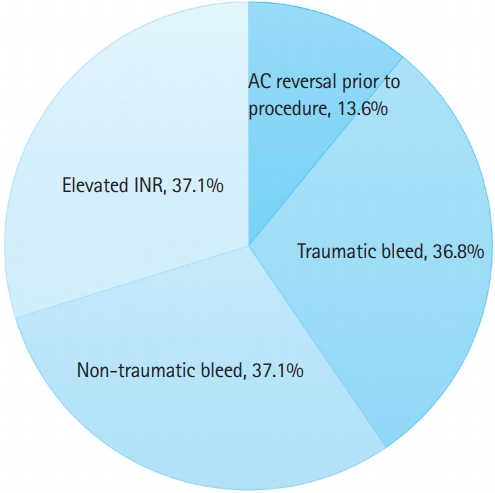
Indications for 4-factor prothrombin complex concentrate. INR, international normalized ratio; AC, anticoagulation.
Concomitant medications of interest to this study included warfarin, aspirin, clopidogrel, and direct oral anticoagulants; data for each was extracted for the patient cohort. The majority of patients were on warfarin therapy (71.1%), followed by aspirin (44.0%), direct oral anticoagulants (15.2%), and clopidogrel (9.2%). Indications for anticoagulation included atrial fibrillation in 63.0% of patients, deep venous thrombosis in 15.8%, pulmonary embolism in 6.5%, prophylaxis for venous thromboembolism in 8.2% and mitral valve replacement in 3.3%. 13.1% were on no anticoagulant prior to 4F-PCC administration. Concurrent treatments for bleeding included vitamin K (58.2%), packed red blood cells (50%), FFP (38%), platelets (26.1%), factor VII (2.2%), factor IX (0.5%), and cryoprecipitate (8.2%). Interventions included surgery in 27.2% and interventional radiology in 8.8% of patients. Patient demographics are outlined in Table 1.
Median (interquartile range) length of hospital stay was 8.4 (4.2–16.9) days. Nine patients (4.9%) developed thrombosis within 90 days of 4F-PCC; four developed deep vein thrombosis, two developed pulmonary emboli, one developed stroke, one developed thrombosis of a stent, and one developed acute thrombus of the left radial artery. In-hospital mortality was 24.5% across the entire patient cohort. Mortality was higher for patients who received 4F-PCC for off-label indications (19.1% on-label mortality vs. 37.7% off-label mortality, P=0.01) (Fig. 2). A comparison of patients treated on- and off-label is presented in Table 2.

Mortality by indication for 4-factor prothrombin complex concentrate. (A) Off-label and (B) on-label use of 4-factor prothrombin complex concentrate.
DISCUSSION
The principal findings of this retrospective chart review of adult patients receiving 4F-PCC in the emergency department were (1) the most common indication for administration was active bleeding, with traumatic and spontaneous bleeds being equally likely; (2) overall mortality rate was 24.6%; (3) there was an approximately two-fold increase in mortality for off-label use compared to on-label use of 4F-PCC; and (4) thromboembolic events (TEE) constituted a small but clinically relevant complication following 4F-PCC.
Reported mortality rates associated with 4F-PCC administration vary widely in the literature and may be attributed to the disease specific state of these cohort analyses. Huang et al. [14], for example, describe a mortality rate of 51.6% in patients with liver pathologies compared to 18.5% in non-liver disease patients. Fischer et al. [15] report a lower in-hospital mortality of 25% following reversal of warfarin-associated intracranial hemorrhage with 3F-PCC or 4F-PCC, with more deaths in the 4F-PCC subset. We comparably demonstrate that 75.4% of our patient cohort, comprised solely of 4F-PCC recipients, survived to hospital discharge. In contrast, Berndtson et al. [16] describe an in-hospital mortality rate of 0% and no thromboembolic complications in patients receiving 4F-PCC for reversal of warfarin-induced coagulopathy, though their sample size was relatively small at 26 patients.
While population-specific findings are useful for determining safety and efficacy data for a respective cohort of patients, these analyses fail to comprehensively demonstrate how 4F-PCC is being utilized by providers in practice, hence limiting our understanding of the potential scope of indications and associated outcomes for this relatively novel agent. Here, we characterize 4F-PCC use across all adult patients, irrespective of existing patient comorbidities, in the emergency department setting, which often serves as the initial point-of-contact for patients with acute bleeds or requiring urgent surgery.
Even though 4F-PCC has been marketed in Europe as Beriplex P/N (CSL Behring, Marburg, Germany) for a number of years, it only gained US Food and Drug Administration approval in 2013 for reversal in the setting of an acute major bleed or urgent surgical or invasive procedure [17]. This study evaluates mortality rates for patients receiving 4F-PCC for on- versus off-label indications, including traumatic and spontaneous bleeds outside the scope of a “major bleed.” Acute major bleeding was defined by Sarode et al. [8] in their phase IIIb study of 4F-PCC as one of the following: life-threatening or potentially life-threatening (according to the treating physician); acute bleeding associated with a fall in hemoglobin ≥2 g/dL; and bleeding requiring blood product transfusion. We analyzed patient charts to correlate these parameters with time of 4F-PCC administration and found inconsistent reporting, necessitating future exploration of whether and how bleed severity is being considered by physicians in their decision-making to utilize this drug for reversal. In our study, the most common bleeding sites were intracranial and gastrointestinal, occurring in over one half of those presenting with an acute bleed. Interestingly, we found that amongst those presenting with no bleed, roughly half were reversed for the off-label indication of an elevated INR without an impending urgent surgical or invasive procedure.
To our knowledge, this is the first study to describe a nearly two-fold increase in mortality amongst patients receiving 4F-PCC for off-label indications. It is likely that those who received 4F-PCC off-label were administered the drug once other therapeutic interventions had been exhausted; these patients were probably sicker and more likely to die. Additionally, a subset of our patients underwent surgery (27.2%) and interventional radiology (8.8%). These procedures carry their own inherent complication risk, including mortality, potentially adversely affecting patient outcomes. Future analyses stratifying patients per comorbidity would allow us to appreciate risk factors for 4F-PCC-associated mortality. There remains a need for randomized controlled trials targeted towards these subpopulations to inform potential expansion of indications for 4F-PCC in the clinical setting.
TEE have been described as a serious concern following 4F-PCC administration. Sin et al. [18] report a rate of 11.8% of TEE within 14 days following 4F-PCC administration, with significant risk factors including heparin-induced thrombocytopenia, major surgery within 14 days, and the presence of greater than six thrombotic risk factors. Joseph et al. [19] similarly describe TEE in 6.2% of patients, with factor V Leiden or antiphospholipid syndrome being predictive on multivariate analysis. Our findings echo those in the literature. While we do not elucidate risk factors for TEE, we describe TEE incidence of 4.9% within 90 days of 4F-PCC, with deep vein thrombosis constituting approximately half of these events. A prior case report of 3F-PCC has described new limb-threatening deep vein thrombosis as a complication of 3F-PCC, and our findings suggest that this complication may also develop in 4F-PCC treated patients, likely due to shared mechanisms related to factor replacement in the coagulation cascade as well as patients’ underlying thrombotic risk [20]. It has been postulated that the higher factor VII content of 4F-PCC compared to 3F-PCC puts patients at higher risk for clot development [1].
Additional complications associated with 4F-PCC include INR rebound and rebleeding, and it has been suggested that intravenous vitamin K be given if a sustained reduction in INR is desired [18]. Indeed, we report that vitamin K was the most common concurrent treatment for bleeding, followed by packed red blood cells, FFP, and platelets. Most patients in our study were on warfarin therapy, which was expected as 4F-PCC is specifically indicated for warfarin reversal. Approximately one-tenth of our cohort was on a direct oral anticoagulant prior to 4F-PCC administration. Studies suggest that while 4F-PCC may be a promising candidate for the reversal of rivaroxaban and apixaban, data supporting this clinical utilization is lacking [21]. Direct oral anticoagulant reversal remains a potential area for expansion of 4F-PCC clinical application.
Our study has several limitations. It is a single-institution retrospective chart review at an academic suburban center. Findings in our study may not be generalizable to patient populations who reside elsewhere. While our findings point to higher rates of mortality for patients receiving 4F-PCC for an off-label indication, our study is not designed to establish a causal relationship between these variables.
As this is a descriptive study, we did not control for potential confounders, including existing comorbidities or the concomitant use of anticoagulant or antiplatelet drugs, which could have affected patient response to 4F-PCC and subsequent morbidity and mortality outcomes. Additionally, patients who were discharged from our emergency department following 4F-PCC administration and presented for thrombosis elsewhere were outside the scope of our electronic medical records and limited our ability to capture all adverse events following 4F-PCC administration.
This study assumes that the data points of interest were comprehensively and routinely recorded by healthcare practitioners in patient charts. During the review, inconsistencies were noted in how units of blood products were recorded both within a single chart and across multiple charts. We attempted to minimize discrepancies in data extraction and interpretation by appointing three independent reviewers, with inconsistencies resolved via discussion.
In conclusion, 4F-PCC is being utilized for indications other than the reversal of warfarin-induced coagulopathy, with higher mortality rates noted for the subset of patients receiving the agent for off-label indications. Further investigation in a controlled clinical setting is warranted to determine the efficacy and safety of 4F-PCC for such indications.
Notes
Adam J. Singer serves as Editor-in-Chief of Clinical and Experimental Emergency Medicine, but had no role in the decision to publish this article. AJS is a consultant and speaker for Alexion, Janssen, Pfizer, and BMS.
CORRECTION
This article was corrected on September 18, 2023, to fix errors in Fig. 2B.
References
Article information Continued
Notes
Capsule Summary
What is already known
Four-factor prothrombin complex concentrate is US Food and Drug Administration approved for treating major bleeding associated with warfarin use.
What is new in the current study
We found that off-label use of 4-factor prothrombin complex concentrate is not uncommon and that mortality is higher with off-label use.