AbstractObjectiveWe evaluated the effect of rapid influenza diagnostic tests (RIDTs) on patient management in an emergency department for 3 years after 2009, and also identified factors associated with the choice of treatment for patients with influenza-like illnesses.
MethodsThe study period consisted of three influenza epidemic seasons. Patients older than 15 years who underwent RIDTs in the emergency department and were then discharged without admission were included.
ResultsA total of 453 patients were enrolled, 114 of whom had positive RIDT results and 339 had negative results. Antiviral medication was prescribed to 103 patients (90.4%) who had positive RIDT results, while 1 patient (0.3%) who tested negative was treated with antivirals (P<0.001). Conservative care was administered to 11 RIDT-positive patients (9.6%) and 244 RIDT-negative patients (72.0%) (P<0.001). Symptom onset in less than 48 hours, being older than 65 years, and the presence of comorbidities were not associated with the administration of antiviral therapy.
ConclusionRIDT results had a critical effect on physician decision-making regarding antiviral treatment for patients with influenza-like illnesses in the emergency department. However, symptom onset in less than 48 hours, old age, and comorbidities, which are all indications for antiviral therapy, were not found to influence the administration of antiviral treatment.
INTRODUCTIONThe influenza virus, which is prevalent each year due to antigenic shift and drift, is an important cause of acute respiratory infection in all age groups [1]. In 2009, due to an instance of antigenic shift, influenza A (H1N1) caused a worldwide pandemic outbreak in 214 countries, resulting in more than 18,306 deaths [2,3]. After the emergence of influenza A (H1N1), a marked increase was noted in the number of patients visiting the emergency department (ED) with suspected influenza infections.
It has become increasingly important to detect influenza rapidly in the ED, as appropriate treatment during the early stages of clinical symptoms can prevent complications and the spread of infection. Various diagnostic tools are used to detect influenza in clinical settings, although most definitive tests require a significant amount of time [4,5]. In contrast, rapid influenza diagnostic tests (RIDTs) are relatively simple, inexpensive, and convenient tests that provide point-of-care results in 10 to 30 minutes. RIDTs are now preferred over other tests in the ED, as emergency physicians are under pressure to rapidly diagnose influenza. However, the implementation of RIDTs poses some concerns. In particular, it has been reported that a certain degree of caution is necessary in interpreting the results of RIDTs and using them in treatment planning for patients with influenza-like illnesses (ILIs) [6-8].
Little research on the effects of RIDTs on the management of adult patients with ILIs in the ED has been conducted after 2009. In this study, we investigated the effect of RIDTs on the management of patients presenting with ILIs in the ED for 3 years after 2009. We also identified whether patients with ILIs in the ED are treated according to treatment guidelines.
METHODSStudy design and settingThis study was conducted in the ED of a tertiary suburban academic hospital serving 45,000 persons annually. Data on the enrolled patients were collected retrospectively from their electronic medical records by an experienced research nurse, and we performed an analysis of RIDT results and treatment modalities.
MethodsThe study period consisted of 3 epidemic influenza seasons: October 2010 to March 2011, October 2011 to March 2012, and October 2012 to March 2013. RIDTs were performed at the discretion of the emergency physician without written guidance in patients who showed flu-like symptoms, respiratory symptoms, or a fever of uncertain cause during the study period. RIDTs were performed using nasopharyngeal swabs with the Genedia influenza Ag (Green Cross Corp., Yongin, Korea) in the early period of the study (2010 to 2011) and the SD Bioline rapid influenza kit (Standard Diagnostics Inc., Yongin, Korea) from 2011 to 2013 [9].
All patients older than 15 years who underwent an RIDT in the ED before being discharged without admission were included in this study, while patients who were admitted, were transferred, or died were excluded. Through a retrospective chart review by an attending physician in the ED, patients whose final diagnosis was unlikely to be an acute respiratory tract infection were excluded from the study. Data on the following variables were collected: vital signs, comorbidities, symptoms, onset of symptoms, performance of chest X-rays, complete blood count, urinalysis, blood cultures, total time spent in the ED, and RIDT results. The treatment modality was classified as antiviral, antibiotic, or conservative treatment.
ILIs were defined as a fever greater than 37.8┬░C with one or more respiratory symptoms in the absence of other causes [10]. We defined comorbidities as chronic medical conditions, such as asthma, chronic lung disease, heart disease, kidney disorders, endocrine disorders, a weakened immune system, and other similar conditions [10].
Statistical analysesAll continuous data were non-parametrically distributed and are presented as mean values with standard deviation. StudentŌĆÖs t-test for continuous variables was applied for comparisons between the two groups. Categorical variables are presented as frequencies with percentages, and were compared using the chi-squared or Fisher exact tests. Multinomial logistic regression was used to identify factors affecting the choice of treatment, using conservative management as the reference group. Data were analyzed using IBM SPSS Statistics ver. 20 (IBM Corp., Armonk, NY, USA). P<0.05 was considered to indicate statistical significance.
RESULTSIn the three time intervals contained within the study period, 3008 patients were tested for influenza using RIDTs, 2,555 of whom were excluded from this study. Among the excluded patients, 1,190 were younger than 15 years and 1,123 were admitted, were transferred, or died. Among the 695 eligible patients, 242 were excluded due to a lack of ILI symptoms. Finally, a total of 453 patients were included in the study (Fig. 1). Among the 453 study patients, 114 had positive RIDT results, while 339 had negative results.
The study patients were divided into RIDT-positive and -negative groups. The two groups did not differ significantly in terms of demographics, such as sex, age, comorbidities, and time since the symptom onset (Table 1). Results of additional examinations, such as chest X-rays, blood tests, urine tests, and cultures, did not significantly differ between the two groups (Table 2). No significant difference was found between the two groups regarding the length of stay in the ED (P=0.250). Antiviral medication was prescribed to 103 patients (90.4%) who had positive RIDT results, but only 1 patient (0.3%) in the RIDT-negative group was treated with antivirals (P<0.001). Antibiotics were not prescribed to any RIDT-positive patients (0%) and were prescribed to 94 RIDT-negative patients (27.7%). Conservative treatment was administered to 11 RIDT-positive patients (9.6%) and 244 RIDT-negative patients (72.0%) (P<0.001).
Multinomial logistic analysis with conservative care as the reference group was used to investigate the association between indications for antiviral and actual treatment (Table 3). Presentation less than 48 hours after the onset of symptoms, being older than 65 years, and the presence of comorbidities, which are all indications for antiviral treatment, were included in the multinomial regression model as covariates. These variables were not found to be associated with antiviral therapy (adjusted odds ratio [aOR], 0.338; 95% confidence interval [CI], 0.074 to 2.023; aOR, 2.089; 95% CI, 0.067 to 64.729; aOR, 3.729; 95% CI, 0.268 to 51.947; respectively). In contrast, old age and presentation less than 48 hours after the onset of symptoms were significantly associated with antibiotic treatment (aOR, 2.096; 95% CI, 1.010 to 4.351; aOR, 1.492; 95% CI, 0.788 to 2.825; respectively).
DISCUSSIONIn the present retrospective study, we found that most patients with positive RIDT results received antiviral treatment, while only 9.6% of RIDT-positive patients received conservative care. Additionally, multinomial logistic regression showed that elapsed time since symptom onset, age, and the presence of comorbidities were not associated with antiviral therapy in patients with ILIs. These results indicate that emergency physicians were completely reliant on RIDT results alone for decision-making about patient management.
Most healthy people infected with the influenza virus recover spontaneously without special treatment, which is referred to as conservative care, but some patients experience influenza-associated complications, such as pneumonia, which is associated with a considerable socioeconomic burden [11]. The timely detection of influenza infections and initiation of treatment reduce the burden associated with the disease, and also prevent the spread of infection [12-15]. However, diagnosing influenza based on clinical symptoms is challenging because many other respiratory viruses produce similar symptoms [16]. Therefore, appropriate laboratory tests in patients with a suspicious clinical presentation are mandatory to ensure the early and accurate diagnosis of influenza.
Most definitive influenza testing methods require a significant amount of time, as well as specialized equipment and trained operators. RIDTs, in contrast, are simple tests, require minimal training, and the results are confirmed immediately [4]. Currently, the use of RIDTs is recommended by the World Health Organization [5]. The use of appropriate RIDTs during influenza epidemics may facilitate the appropriate prescription of antivirals, as well as potentially reduce the number of additional tests ordered in the ED, the amount of antibiotics prescribed, and the length of hospital stays [17-20].
An RIDT alone, however, is insufficient to confirm influenza infection. Sensitivity and specificity analyses have been performed on various RIDTs, and a meta-analysis has reported a sensitivity of 62.3% (95% CI, 57.9% to 66.6%) compared with reverse transcription polymerase chain reaction [21-24]. Additionally, even for a single RIDT, different populations and different samples can lead to different sensitivities [15,25]. Caution must be heeded when interpreting RIDT results due to their inconsistent accuracy; while influenza can be considered, it cannot be excluded based on RIDT results alone, and emergency physicians or primary physicians caring for patients with ILIs should not base decisions regarding antiviral prescription solely on RIDT results.
Most patients with negative RIDT results in this study did not receive antiviral treatment. Negative RIDT results have a reasonable likelihood of being false negative. If an RIDT yields a negative result in a patient with an ILI who is old or has a high-risk medical condition, a clinician should not exclude influenza and defer the initiation of antiviral therapy in accordance with his or her clinical judgment. Based on the limitations of laboratory diagnostic tests, even if RIDT results are negative, recent guidelines for influenza management recommend that antivirals be administered as treatment in high-risk patients who have a higher risk of serious influenza complications [13,26].
Most otherwise healthy people who become sick with influenza do not require antiviral drugs, as most influenza infections are self-limiting. Antiviral therapy is most effective when administered early in the course of illness, within 48 hours after the onset of symptoms. Additionally, antiviral agents are associated with frequent side effects. The risk-to-benefit ratio of medications considering the time elapsed after symptom onset, in addition to the RIDT results, should be considered in the decision to initiate antiviral therapy.
Additionally, unlike past studies, our analysis did not reveal a difference in the length of stay in the ED or the performance of additional testing according to RIDT results [18,20]. This is most likely due to the fact that this study involved only adult patients discharged from the ED, in whom the symptoms were mild to moderate.
This retrospective study has several limitations. First, it was based on 3 years of medical records from one hospital, which may not be representative of all Korean hospitals, thereby reducing the generalizability of our findings. Second, this study may have some degree of selection bias, as it included patients who underwent RIDTs at the emergency physicianŌĆÖs discretion. Another limitation is that relatively few patients were enrolled in the study. Nonetheless, significant differences were found between the RIDT-positive and RIDT-negative groups in terms of treatment strategies. Finally, we did not consider local influenza prevalence during these seasons. RIDTs are of limited use when the local influenza prevalence is low [14]. The prevalence of influenza varies between and within seasons, which can affect how physicians interpret RIDT results.
The present study suggests that RIDT results had a major effect on physician decision-making regarding the initiation of antiviral treatment for patients with ILIs in the ED. However, symptom onset within 48 hours of presentation, old age, and the presence of comorbidities were not found to influence the use of antivirals. Although RIDTs have several advantages, careful clinical evaluations in conjunction with RIDT results are necessary to ensure that patients with ILIs receive appropriate clinical care.
REFERENCES1. Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev 1992; 56:152-79.
2. Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team, Dawood FS, Jain S, et al. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009; 360:2605-15.
3. World Health Organization. Pandemic (H1N1) 2009: update 100 [Internet]. Geneva: World Health Organization; 2010 [cited 2017 Dec 30]. Available from: http://www.who.int/csr/don/2010_05_14/en/.
5. World Health Organization. WHO recommendations on the use of rapid testing for influenza diagnosis [Internet]. Geneva: World Health Organization; 2005 [cited 2017 Dec 30]. Available from: http://www.who.int/influenza/resources/documents/RapidTestInfluenza_WebVersion.pdf?ua=1. Accessed Dec 30, 2017.
6. Vasoo S, Stevens J, Singh K. Rapid antigen tests for diagnosis of pandemic (Swine) influenza A/H1N1. Clin Infect Dis 2009; 49:1090-3.
7. Louie JK, Guevara H, Boston E, et al. Rapid influenza antigen test for diagnosis of pandemic (H1N1) 2009. Emerg Infect Dis 2010; 16:824-6.
8. Choi WS, Noh JY, Huh JY, et al. The clinical usefulness of the SD Bioline Influenza Antigen Test for detecting the 2009 influenza A (H1N1) virus. Yonsei Med J 2011; 52:683-5.
9. Cho CH, Woo MK, Kim JY, et al. Evaluation of five rapid diagnostic kits for influenza A/B virus. J Virol Methods 2013; 187:51-6.
10. Centers for Disease Control and Prevention. People at high risk of developing flu-related complications [Internet]. Atlanta, GA: Centers for Disease Control and Prevention; 2016 [cited 2017 Dec 30]. Available from: https://www.cdc.gov/flu/about/disease/high_risk.htm.
11. Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004; 292:1333-40.
12. Low D. Reducing antibiotic use in influenza: challenges and rewards. Clin Microbiol Infect 2008; 14:298-306.
13. Choi WS, Lee J, Lee HY, et al. Clinical practice guideline for antiviral treatment and chemoprophylaxis of seasonal influenza. Infect Chemother 2012; 44:233-49.
14. Grijalva CG, Poehling KA, Edwards KM, et al. Accuracy and interpretation of rapid influenza tests in children. Pediatrics 2007; 119:e6-11.
15. Centers for Disease Control and Prevention. Influenza sings and symptoms and the role of laboratory diagnostics [Internet]. Atlanta, GA: Centers for Disease Control and Prevention; 2016 [cited 2017 Dec 30]. Available from: http://www.cdc.gov/flu/professionals/diagnosis/labrolesprocedures.htm.
16. Call SA, Vollenweider MA, Hornung CA, Simel DL, McKinney WP. Does this patient have influenza? JAMA 2005; 293:987-97.
17. Noyola DE, Demmler GJ. Effect of rapid diagnosis on management of influenza A infections. Pediatr Infect Dis J 2000; 19:303-7.
18. Bonner AB, Monroe KW, Talley LI, Klasner AE, Kimberlin DW. Impact of the rapid diagnosis of influenza on physician decision-making and patient management in the pediatric emergency department: results of a randomized, prospective, controlled trial. Pediatrics 2003; 112:363-7.
19. Abanses JC, Dowd MD, Simon SD, Sharma V. Impact of rapid influenza testing at triage on management of febrile infants and young children. Pediatr Emerg Care 2006; 22:145-9.
20. Iyer SB, Gerber MA, Pomerantz WJ, Mortensen JE, Ruddy RM. Effect of point-of-care influenza testing on management of febrile children. Acad Emerg Med 2006; 13:1259-68.
21. Babin SM, Hsieh YH, Rothman RE, Gaydos CA. A meta-analysis of point-of-care laboratory tests in the diagnosis of novel 2009 swine-lineage pandemic influenza A (H1N1). Diagn Microbiol Infect Dis 2011; 69:410-8.
22. Faix DJ, Sherman SS, Waterman SH. Rapid-test sensitivity for novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009; 361:728-9.
23. Jacobus CH, Raja AS. How accurate are rapid influenza diagnostic tests? Ann Emerg Med 2013; 61:89-90.
Fig.┬Ā1.Schematic flowchart of patient inclusion in the study. RIDT, rapid influenza diagnostic test; ILI, influenza-like illness. 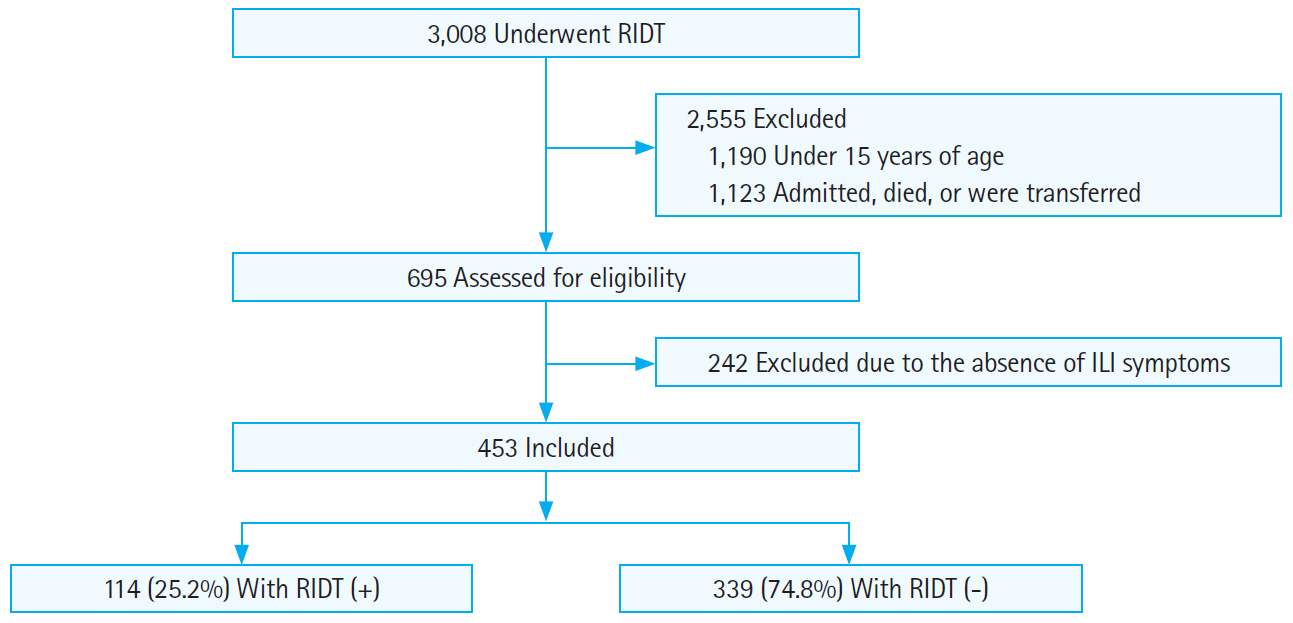
Table┬Ā1.Baseline characteristics of study patients
Table┬Ā2.Outcomes of patient management in the ED according to RIDT results Table┬Ā3.Factors associated with the treatment of study patients, determined using a multinomial logistic regression model with conservative care as the reference group
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||